-
Posts
23534 -
Joined
-
Days Won
193
Everything posted by SteamyTea
-
What make is that? Would do most of my daytime loads.
-
I think that boat has sailed
-
It does take that into account, the ⁰C/hour figures are decrement. This notional 12 hours is of no use in the UK, our latitude is too high.
-
Yes, this probably has the greatest effect, along with uncontrolled/un designed solar gain and ventilation. I think the main thing is that if someone says 'we are building heavyweight because it stops temperature variation', they need to be made to stop and think. If someone says 'we are building heavyweight because we can't find a carpenter' then that is sensible.
-
Oh dear, you still falling for that then. In economics there is a term 'competitive advantage'. The UK is anything but competitive in manufacturing, mainly why we ship our designs out and let others manufacture them. The UK has one of the worse productivity levels of any industrialised nation. Making food and consumer 'stuff' in the UK is wasteful, why we have changed to a service economy, we are good at that, though we are slipping down the rankings fast. Shipping of good is such a tiny part of the cost and the emissions that it is often not able to be measured at the component level. If you drive to a local farm shop you are almost certainly doing more environmental damage than going to the local supermarket, even if that local supermarket is further away. Getting goods delivered to your door is the best option, though that does raise question about how we treat our 'gig' economy workers. Here is an example for you about how hopeless we are. The Bacon company Tulip has a processing plant in Cornwall, it is about 60 miles from Devon, 100 from Exeter. Cornwall is not a pig rearing area, the weather is wrong for it. So they ship carcases in, process them, then ship them out, all by trucks (if you ever see an orange CBT lorry, chances are it is full of bacon). So we rear hundreds of miles from the processing plant, then ship them back, hundreds of miles, to the population centres i.e. Bristol, Birmingham, Manchester, London, Brighton, Hull (closer to Holland than Cornwall) etc. The thing is, this is still more economical, on all levels of sustainability, than a local farmer raising a pig, taking it off to market, then bringing it back as processed meat and selling it to a local. We live in a globalised economy, we have since the East India Trading Company was battling on the high seas, the reason we trade is because it is more efficient, and these days, less damaging. There is a saying that you cannot have 'quick, cheap and good', well you can, you just get the people who have invested in the resources to do the work. They tend to work in integrated manufacturing systems, not alone.
-
Why do you do that?
-
@joe90 There are many reasons for choosing building materials, two of the biggest are availability and local skills. What should not be happening is nonsense about how heavy weight materials thermally stabilise a building, that only happens at the very extremes i.e. a deep cave or a single skin tent. And even then, different inputs are interacting differently. Solid masonry walls are not much different in rigidity than properly designed steel or timber walls. The key point there is 'properly designed'. If all design was equal, we would still be driving Austin Allegros.
-
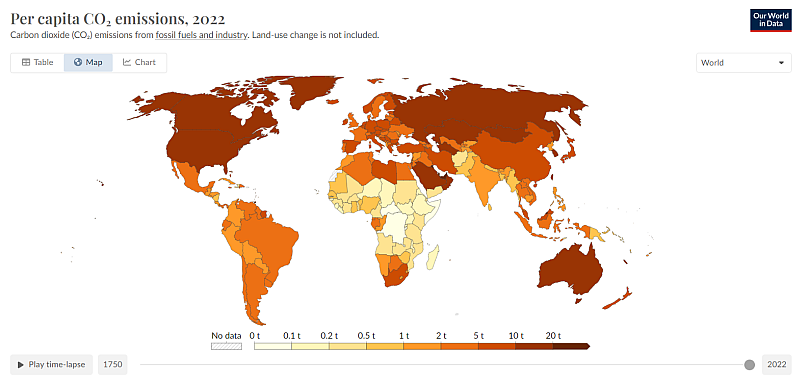
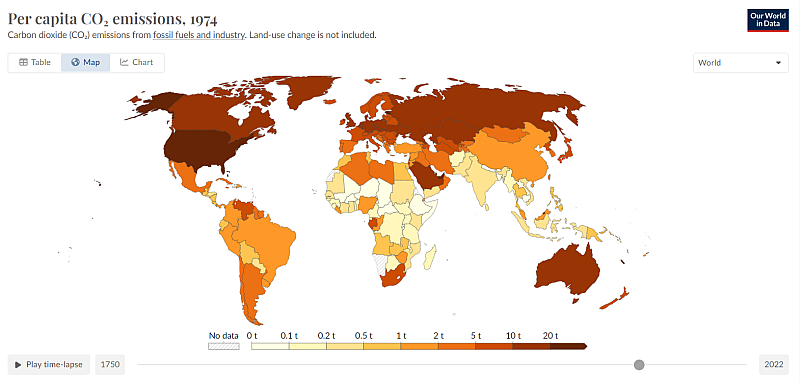
You are telling someone that studied this 2 decades ago, why we have the terms embodied energy and embodied carbon, it is so we can easily compare different items made in different places. Per Capita CO2 emissions is the best way to see the overall picture.
-
Can you elaborate what you think we should be doing to 'protect ourselves'. When it comes to new electrical generation, wind and solar are the cheapest forms, carrying on with thermal plant technology will cost more. I don't think we need to be too hard on the Chinese, they are not much worse than us. And going back 50 years. What I find more interesting is that the historically bad polluters are still the bad polluters.
-
Yes, the marketplace has changed over the last few years. Car dealerships are really selling finance.
-
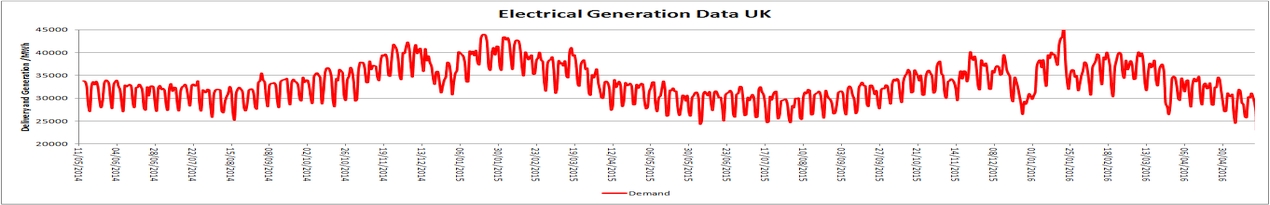
As many of you know, I have been sceptical that adding extra mass to a building will stabilise the temperature. This basically comes from when I studied this back in 2008. Just to reconfirm my suspicions that regardless of construction type, the air in a building will react more to external inputs than internal inputs i.e. solar gain, ventilation and a heating system versus thick stone, brick or concrete walls. To retest this idea, I worked out the amount of energy that is needed to change the air in one of my rooms, then calculated how much water is needed to match it. Then I started measuring temperatures over the last few days. Now I can bore you all with very detailed statistics, but it boils down to what I showed 16 years ago, it basically makes no difference. The headline figures are that the mean air temperature was 16.7°C with a range of 6.2°C for the air in the room, 6 litres of water, which needs the exact same amount of energy to change by 1°C had a mean temperature of 16°C and a range of 25°C and double that amount, 12 litres, had a mean temperature of 16.3°C and a range of 2.9°C. The bigger range, which some will interpreted as instability is caused by natural air changes and heating input (I have storage heating, so does not modulate like a properly set up combustion or heat pump system). When looking at the more stable stable times between 10 AM and 3 PM (when I am usually out) the mean temperatures are, for the room 17°C (range 0.1°C), 6 litres 16.1°C (range 0°C), 12 litres of water 16.4°C (range 0°C). So allowing for instrument accuracy, about the same range, but the masses are actually cooling the air. When I looked at the rate if change in an hour, all three were the same at -0.1°C/hour when cooling, and when warming, the air reacted faster at 0.3°C/hour but only over the time the energy is being inputted (two hours in my case). The water masses are equal at 0.1°C/hour. So the air responded a bit faster overall, but not much over 24 hours. All the above confirms what I researched 16 years, adding mass to a building will make the mean temperature lower. If you want to stop overheating, change the window design, no need to fill the walls with concrete, brick or block. That is barking up the wrong tree.
-
As most people do not own a car these days, but have it on a lease of some sort, the market could be flooded because there were a lot leased out in 2020. Like most of these things, they are a lot more complicated than they seem at first. Mind you, I would be a bit miffed if Ford sold me an electric Mustang and then would not take it in part exchange.
-
I think you will find it is a bit more complicated than just asking the question. A lot more details is needed. But here is some light reading https://www.structuralbasics.com/flat-roof-beam-design/ https://www.structuralbasics.com/timber-truss-roof/ https://www.structuralbasics.com/rafter-roof-design/ https://www.structuralbasics.com/purlin-roof-design/
-

Ceiling Speakers Recommendations
SteamyTea replied to richo106's topic in Networks, AV, Security & Automation
What a cop out. Idle (expletive deleted)er -

Ceiling Speakers Recommendations
SteamyTea replied to richo106's topic in Networks, AV, Security & Automation
Or you could do a write up for everyone to benefit. -
Below is a letter in this weeks comic. Weather calamities are climate disasters Published 21 February 2024 From Michael Paine, Sydney, Australia Your view on whether the world has already warmed by 1.5°C refers to extreme weather events. The resulting human tragedies are often referred to as “natural disasters” by politicians and the media. But the causes are becoming less and less natural. Let’s start calling them what they are – “climate disasters” (Leader, 10 February). Here is the Leader. Forget what long-lived sea sponges say, focus on reaching net-zero According to data from Caribbean sponges, the world passed 1.5°C of warming a decade ago, but debating these arbitrary limits distracts from the bigger picture 7 February 2024 ERWIN MASCARINAS/AFP via Getty Images IT’S not often that global news is garnered from a sponge. But that is what we find ourselves debating this week. Researchers studying long-lived sea sponges to track the average global surface temperature over 300 years have made a startling discovery: when delegates at the Paris climate conference in 2015 agreed to aim to limit warming to 1.5°C above pre-industrial levels, it may have already been too late. According to data from the sponges, the world was already more than 1.5°C hotter as a result of the burning of fossil fuels. This conclusion is controversial. A key issue is that this species of sponge lives only in the Caribbean. Even if its skeleton does accurately record past temperature changes, extrapolating globally from one region is questionable. However, the idea that fossil fuels have warmed the planet somewhat more than the official figures suggest is plausible. But debates about whether we have passed 1.5°C distract from the bigger picture. What really matters is that the current level of global warming is already dangerously high, and that further warming will be even more disastrous. We are already seeing ever more extreme weather events around the world, with record-breaking heatwaves, floods and droughts. These extremes are causing increasingly serious knock-on effects. They are reducing productivity, damaging crops, driving up food prices and creating political instability. They are destroying homes and reversing hard-won social and economic advances. They are also disrupting trade, with low water levels in Europe and the Panama Canal affecting shipping (see “Drought has hit the Panama Canal hard – can it survive climate change?”). To prevent things from getting much worse, we need to stop the world warming further. Achieving this will require global greenhouse gas emissions to reach net zero. All the focus should therefore be on reducing emissions, not on discussing rather arbitrary temperature limits. And with global emissions still rising, we have an awful lot of work to do.
-
I use this at home. At work, this. It rips my skin off. You can make your own with this, just make a paste with water. Just don't throw it in anyones face, then jump of a Thames bridge or they will ban the sale of it.
-
You could rent a caravan on a holiday park, you can take your own bucket to pee in.
-

Designing with contingency for any future crisis/emergency
SteamyTea replied to saveasteading's topic in Barn Conversions
It is quite interesting how we could build in resilience to our housing stock. I suspect that it is not very expensive to do. Taking away the extreme examples of nuclear war, or regular missile attacks, and thinking about what has happened in the UK over the last decade. Remember the floods in 2013/14 Same year we had 5 storms hit the south west in 7 days, with Hercules being the worse. We had that 2018 'Beast from the East' 2018 and 2022 we had a heat wave In isolation none of them are unique, but because they have happened in a decade, and extreme weather events are becoming more common, and with more unpredictable consequences, we really do need to start planning for at least a few days disruption every year. Those few days disruption may not always be the same, and may not be weather events, remember the 2000 fuel protest. So what can sensibly be done. As I was sitting having a coffee earlier, the RNLI had their chuggers in the cafe. Now I support the RNLI, so felt no need to engage in conversation with them and then say they cannot have more money from me. But it did get me thinking about communications when things go wrong. Mobile phones are great, until they are not connecting. But we have two radio systems that are used a lot in the UK, and most of us never use them, or even think about them. They are the Taxi Radio system and the Marine VHF Radio system. I think the latest Taxi Radios are become centralised, so I am talking about the older sorts. I am not sure how much they cost to buy, and I think that you are meant to have a licence, but in a national emergency, I think you will get away with any qualifications. A decent normal radio is also worth having, especially a wind up one. I am not sure how much is on the short wave these days, it used to be the frequency of choice for ex-pats. The next thing is fresh water. This is very easy and cheap, just get some 30 litre containers and fill them up each week. Allow for 5 litres a day per person for drinking and cooking. Easy. If life is really bad, get one of these they work well enough when I am walking. Food is very easy, tinned and dried. The reason I don't have a freezer is that I have a number of large supermarkets nearby, and they are full of freezers, so I just buy frozen things when I need them, the rest is tinned. Camping stoves are brilliant. I have a tiny 3 kW one, it is ferocious. If you have land, and time/interest, start growing things that you can eat. Pick the high calorie foods, not lettuce. Fruit trees are nice, and apples are full of goodness, and sugar. The more I think about it, the less important space heating is. Wearing thermals is good for keeping the chill off when moving, wrapping up in half decent winter walking clothing will keep you warm, and dry. Get several different types of gloves, large loose ones and tighter thinner ones. It is impossible to open a tin of soup in mittens. You can sleep fully clothed, not compulsory to put the baby doll out fits on. I am sure I will think up more things, most of us will already have most of the stuff we actually need as we tend to be 'out doorsy types'. -
There are small chapels in the mid west of the USA that will welcome you with open arms.
-

Designing with contingency for any future crisis/emergency
SteamyTea replied to saveasteading's topic in Barn Conversions
-
Not sure what that is referring to.
-
Yes, I heard that, but did not take take much notice as I was driving in exceptionally wet weather, not that week's of rainfall has (expletive deleted) all to do with climate change, it is just weather isn't it.
-

Designing with contingency for any future crisis/emergency
SteamyTea replied to saveasteading's topic in Barn Conversions
Actually, a caravan or campervan is a good idea if it can be stored out the way. Does not have to be a working one, just cosy. Boats, by definition, are off grid.